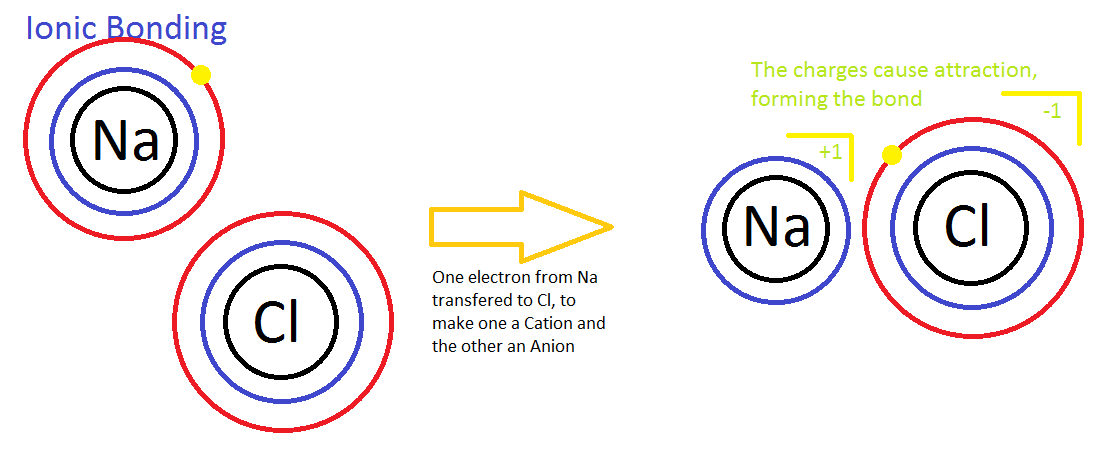
- Ionic Bond: formed by the attraction of oppositely charged atoms
- Formed by the attraction of oppositely charged atoms
- When an atom gains or loses one or more electrons form an Ion
- Cations:
- When a positively charged Ion is attracted to the negatively charge "cathode" in an electrical field
- Anions:
- When a negatively charged ion is attracted to the positively charged "anode"
- Every Ionic Chemical Bond is made from at least 1 Cation and 1 Anion!!
- Formed by the attraction of oppositely charged atoms
- Covalent Bonds: is a result from sharing of electrons between 2 atoms with similar electronegativities
- A single bond represents the sharing of 2 valance electrons (usually from 2 different atoms)
- Multiple covalent bonds are common for certain atoms depending on their valance electrons
- Metallic Bonds: result of electromagnetism/ describes the electrostatic attractive force that occurs between condition electrons and positively charged metal ion
- Delocalization:
- Used to explain conductivity, malleability, and ductility
- No atom in metal sample has a strong hold on its electrons and shares them with neighbors (bonds)
- Greatest number of electrons per atom that participates in metallic bonding, with stronger bonds
- Delocalization:
- Polyatomic:
- Ions that are composed of 2 or more atoms that are linked by covalent bonds
- Still have a net deficient or surplus of electrons resulting in an overall condition
- A metal plus a polyatomic Ion yields an ionic compound